Today was the first time in the COVID daily briefings that Trump has mentioned the 2018 “Right to Try” as the reason for insistence on untested treatments, confabulating them with cures and vaccines.
It is an extension of the RW deregulatory discourse that when it comes to the desperate time (the reasons for DNR do-not-resuscitate orders) for terminally-ill patients, that false hope can be prescribed. Not unlike Terri Shiavo and Bill Frist, desperation can be manipulated for religious effect.
Trump is doing just that, looking for COVID-19 medical miracles that might provide the political miracle for his 2020 campaign. Truly a Hail Mary. He needs poster children, and confusing FDA approval with other expert opinions is appealing to his GOP base.
Between the lines: "There has never been a confrontation in the task force meetings like the one yesterday," said a source familiar with the argument. "People speak up and there's robust debate, but there's never been a confrontation. Yesterday was the first confrontation."
- In response to a request for comment on Axios' reporting, Katie Miller, a spokesperson for the vice president, said: "We don't comment on meetings in the Situation Room."
The bottom line: The way to discuss the drug's potential has become a fraught issue within the Trump administration.
- Most members of the task force support a cautious approach to discussing the drug until it's proven.
- Navarro, on the other hand, is convinced based on his reading that the drug works against the coronavirus and speaks about it enthusiastically.
- Some of Trump's favorite TV hosts, including Fox's Sean Hannity, and friends including Rudy Giuliani, have also been touting the malaria drug for the coronavirus. Trump has made no secret who he sides with.
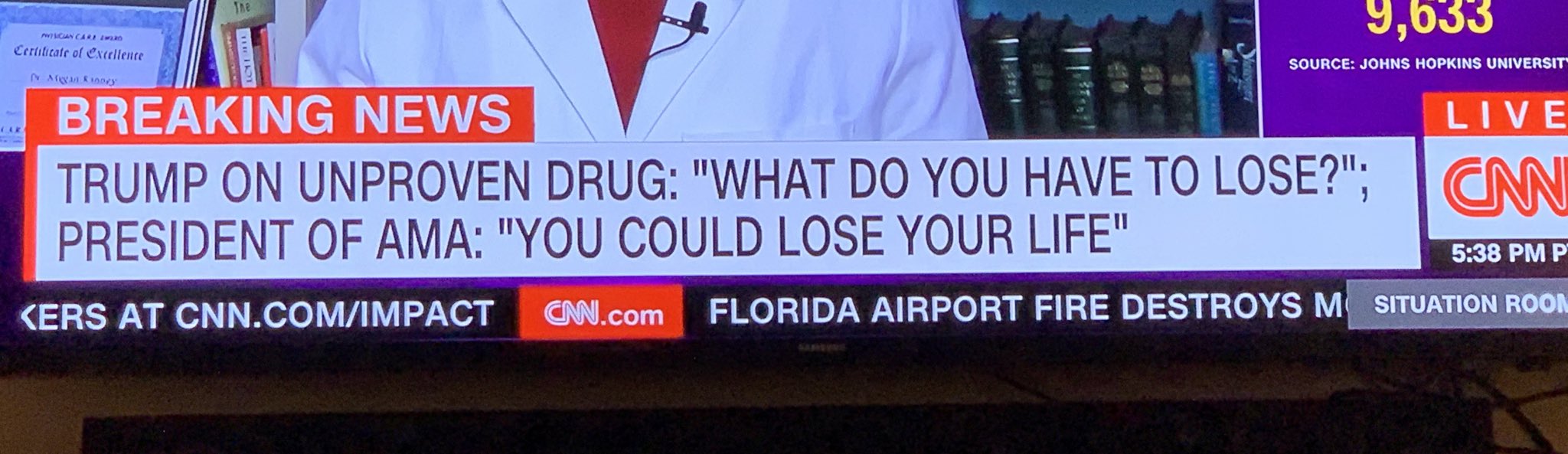
- "What do you have to lose? Take it," the president said in a White House briefing on Saturday. "I really think they should take it. But it's their choice. And it's their doctor's choice or the doctors in the hospital. But hydroxychloroquine. Try it, if you'd like."
Right to try legislation gives terminally ill patients the right to use experimental medications that have not yet been approved by the Food and Drug Administration.
President Trump's push for the passage of right to try nationally builds on the efforts of the libertarian-leaning Goldwater Institute, which has used the broad popularity of the policy to help achieve passage of similar legislation in 38 states, although the state legislation differs substantially from the version now on the president's table.
[...]
Despite these state gains and the policy's growing popularity among states, ethical questions remain about the tangible impact of a federal right to try law on Americans with terminal illnesses. Most notably, a growing body of evidence from policy analysts argues that the legislation would unfortunately accomplish very little change for most patients, and it could actually make it harder to get new drugs approved in the future.
[...]
Ultimately, we believe right to try gives the Trump administration a political win on a popular public policy but will do little to change the status quo for terminally ill patients. Some opponents say, if anything, it could give terminally ill patients false hope for new access to drugs they already can obtain through existing FDA policy.
Even if right to try does simplify federal regulation, patients must still convince a reticent pharmaceutical industry to provide drugs outside of trials, and patients must still bear the costs for these experimental treatments, which is out of reach for almost all Americans.
The right to try legislation will score the president and members on both sides of the aisle in Congress points, but ultimately it will change little for terminally ill patients and their families.
www.cbsnews.com/...


